 
This is the order for the question and the answer for it. 
He returned the electra negativity which is what decreases? Which is your lead's decrease? Here the question is asked, we write the order. When we go through the group of redwoods, we know they looked at the increase in local negative vibes. You're actually electra negative when you're along the period of electro negativity. Which is equal to the electoral negativity of K. R B's electoral negativity was mentioned. He looked at the order of the negative things. So you are prepared? Ready Look Trow was looking at something negative. The electoral order is written in the increasing order of the compound. The program and the script to produce this periodic table.Greetings students. Special thanks to Craig Counterman from MIT who wrote We use rule #4 to decide that NaBr has ionic bonds and that HF has a polar covalent bond in each HF molecule. Hydrogen fluoride (formula = HF EN H = 2.1, EN F = 4.0) has the same DEN. Here is an example: Sodium bromide (formula = NaBr EN Na = 0.9, EN Br = 2.8) has a DEN = 1.9. If only nonmetals are involved, the bond is considered polar covalent. If the DEN is between 1.6 and 2.0 and if a metal is involved, then the bond is considered ionic. What about the gap between 1.6 and 2.0? So, rule #4 is:Ĥ. That, of course, leaves us with a problem. If the DEN is greater than 2.0, then the bond is ionic. If the DEN is between 0.5 and 1.6, the bond is considered polar covalentģ. If the electronegativity difference (usually called DEN) is less than 0.5, then the bond is nonpolar covalent.Ģ. The rule is that when the electronegativity difference is greater than 2.0, the bond is considered ionic.ġ. Substances such as NaCl and MgCl 2 are the usual examples. Ionic: This type of bond occurs when there is complete transfer (between the two atoms) of the electrons in the bond. This last example is about as polar as a bond can get. (Some textbooks or web sites use 1.7.) Obviously there is a wide range in bond polarity, with the difference in a C-Cl bond being 0.5 - considered just barely polar - to the difference the H-O bonds in water being 1.4 and in H-F the difference is 1.9. The typical rule is that bonds with an electronegativity difference less than 1.6 are considered polar. Molecules such as NH 3 and H 2O are the usual examples. It assumes that bonds and lone pairs repel each other, and will arrange. Polar Covalent: This type of bond occurs when there is unequal sharing (between the two atoms) of the electrons in the bond. According to the VSEPR theory, valence shell electron pairs will stay as far. Since the electronegativities of C and S are both 2.5, you have a nonpolar bond. 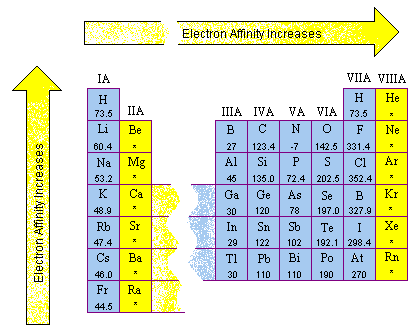
Sometimes a teacher will only use diatomics as examples in lecture and then spring CS 2 as a test question. One interesting example molecule is CS 2. Since textbooks vary, make sure to check with your teacher for the value he/she wants. Textbooks typically use a maximum difference of 0.2 - 0.5 to indicate nonpolar covalent. Molecules such as Cl 2, H 2 and F 2 are the usual examples. Nonpolar Covalent: This type of bond occurs when there is equal sharing (between the two atoms) of the electrons in the bond. Only the absolute difference is important. Calculate the difference between their electronegativity values. What you should do is look only at the two atoms in a given bond. This pattern will help when you are asked to put several bonds in order from most to least ionic without using the values themselves.Įlectronegativity values are useful in determining if a bond is to be classified as nonpolar covalent, polar covalent or ionic. The reverse statement is that the values tend to decrease going down and to the left. When you examine a periodic table, you will find that (excluding the noble gases) the electronegativity values tend to increase as you go to the right and up. Oxygen is the 2nd most electronegative element. Because fluorine is the most electronegative element, the electrons tend to "hang out" more toward the fluorine atom when fluorine is covalently bonded to other atoms. :max_bytes(150000):strip_icc()/chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)
In general electronegativity is the measure of an atom's ability to attract electrons to itself in a covalent bond. Pauling did not assign electronegativities to the noble gasses because they typically do not form covalent bonds. Note that atoms toward the upper right are more electronegative, and those to the lower left are least electronegative. Linus Pauling's electronegativity scale is the most common. There are other ways of measuring electronegativity, such as the Mulliken scale and the Allred-Rochow scale. This table is the Pauling electronegativity scale. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
